 
The Atomic Spectrum should be a pure line Spectrum. The characteristics of the Atomic Spectrum are observed as: Spectral series are crucial in Astronomical Spectroscopy. The visible spectral lines in the hydrogen emission Spectrum are caused by Atomic transitions between distinct Energy levels. The Rydberg formula clearly splits the Atomic Hydrogen emission Spectrum into a number of wavelength-dependent spectral lines. An Electron emits or absorbs light of a specific wavelength as it jumps from one Energy level to the next. When an Electron gets excited from one Energy level to another, it emits or absorbs light of a specific wavelength.Īs an Electron moves between different Energy levels within an Atom, its Spectrum of Electromagnetic radiation is released or absorbed. When an Electron transitions from one Energy level to another, it emits light or photon with a specific wavelength. There are more Energy states in a tom than there are Electrons. In an Atom, Electrons have discrete and some specific energies. Each element has a characteristic Spectrum through which it can easily be recognized. 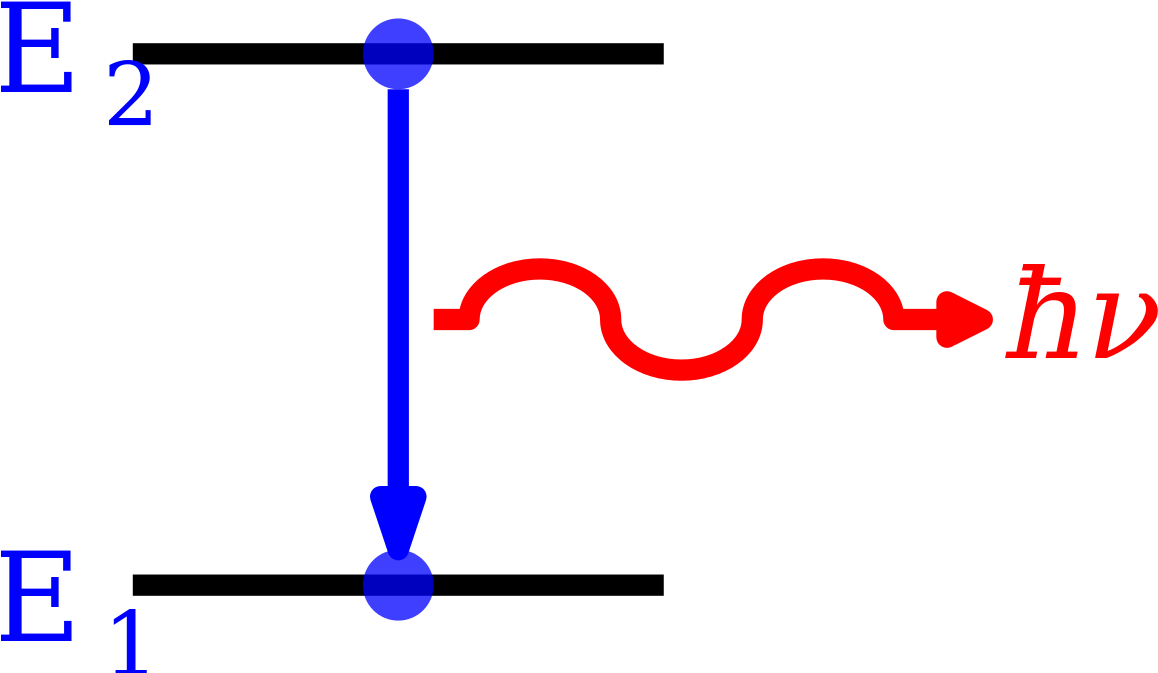
Department of Energy, by the National Aeronautics and Space Administration, by NIST's Standard Reference Data Program (SRDP), and by NIST's SMA Program.The Atomic spectra are defined as the Spectrum of frequencies of electromagnetic radiation emitted or absorbed during transitions of Electrons between Energy levels within an Atom. This database was funded by the Office of Fusion Energy Sciences of the U.S. Online: March 1995 - Last update: October 2022 All rights reserved. NIST reserves the right to charge for these data in the future. Department of Commerce on behalf of the United States. Past Contributors: Haris Kunari, Jean E. Sansonetti, Jeffrey R. Fuhr, Larissa I. Podobedova, Wolfgang L. Wiese, John J. Curry, Gerry R. Dalton, Robert Dragoset, Fun-Chen (Jesse) Jou, William C. Martin, Peter J. Mohr, Arlene Musgrove, Craig J. Sansonetti, and Gloria Wiersma 
Students contributing to data entry: Eric Carpentier, Thomas Carpentier, Amy Zimmerman, Adrian Hamins-Puertolas, Marko Hamins-Puertolas, Anna Sharova, Genevieve Tan NIST ASD Team Principal Developers (Currently Active): Alexander Kramida, Yuri Ralchenko, and Joseph Readerĭata Compilers (Currently Active): Alexander Kramida, Edward B. Salomanĭatabase Developers (Currently Active): Alexander Kramida, Yuri Ralchenko, and Karen Olsen The Data Center is located in the Physical Measurement Laboratory at the National Institute of Standards and Technology (NIST). The Atomic Spectroscopy Data Center has carried out these critical compilations. This database provides access and search capability for NIST critically evaluated data on atomic energy levels, wavelengths, and transition probabilities that are reasonably up-to-date. Help - On-line help in using the database. Synchrotron Ultraviolet Radiation Facility SURF IIIĪSD Interface for Laser Induced Breakdown Spectroscopy (LIBS)Īdditional information about the database may be obtained through the following links:Ītomic Spectroscopy Intro - Outlines basic atomic physics concepts, explains terminology and notation.ĪSD Intro & Contents - Introduction to and contents of the Atomic Spectra Database.īibliography - Bibliography of data sources used for this database.Radiopharmaceutical Standardization Laboratory.Calibrations Customer Survey (external link). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
